This 2018 review combines a comprehensive summary of brain aging with a brief overview of the evidence linking metabolic patterns to acceleration or suppression of brain pathology.
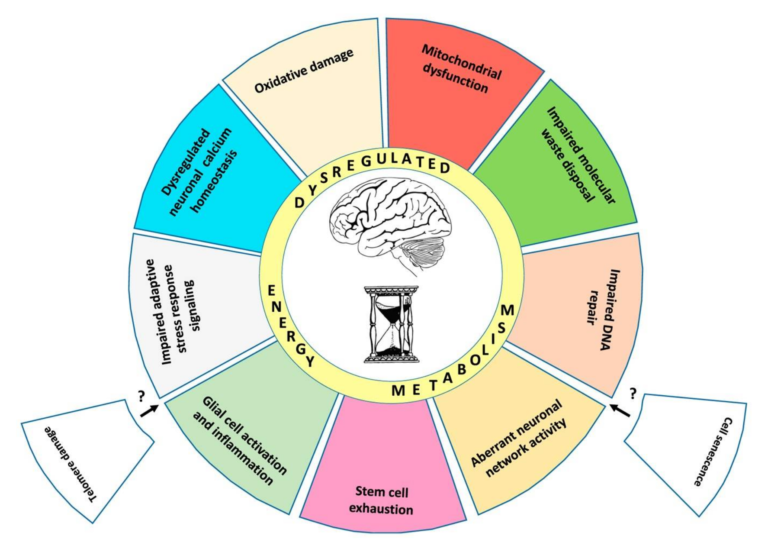
The authors outline 10 “established hallmarks” of brain aging, presented in Figure 1 below.
Briefly:
- Mitochondrial dysfunction – Mitochondria generate the ATP required for neuronal function, maintenance, and repair while also supporting various signaling pathways. Aging is associated with structural and functional defects in the mitochondria, including membrane breakdown and impaired electron transport chain function (1).
- Oxidative damage – Aging leads to the progressive damage of brain proteins when generation of reactive oxygen species (ROS) exceeds what can be cleared by antioxidant defenses. Animal studies have shown that impairments in oxidative defense mechanisms alone can trigger the symptoms of brain aging (2).
- Impaired lysosome and proteasome function – Neurons, unlike most cells, generally do not replicate; instead, they maintain function by removing and replacing damaged molecules. Lysosomes and proteasomes manage this process; animal research indicates the activity of these systems is diminished in the aging brain, and pharmaceutical upregulation of these same functions can reverse neurodegeneration (3).
- Dysregulation of neuronal calcium homeostasis – Calcium ions support the function and adaptation of the neuronal network, and calcium dysregulation is seen in aging brains. A small number of rat studies have indicated restoration of normal calcium levels ameliorates cognitive deficits (4).
- Compromised adaptive cellular responses – Neuronal pathways for responding to stress and activating cellular defense mechanisms, many of which are triggered by changes in ATP and reactive oxygen species levels, are impaired in aging, increasing stress vulnerability (5).
- Aberrant neuronal network activity – Aging is associated with decreased neural network function and structural integrity. The function of the default mode network (DMN), which represents the function of the brain in the absence of deliberate activity, is particularly disordered in the aging and cognitively impaired (6).
- Impaired DNA repair – DNA is continually damaged and repaired as a consequence of normal cellular function, with increases in this damage occurring after synaptic activity. Decreased DNA repair protein activity is seen in older mice, and multiple premature aging syndromes (Cockaye syndrome, Werner syndrome, and ataxia telangiectasia) are caused by mutations in DNA repair processes (7).
- Inflammation – Inflammation levels are increased both in the aging brain and in the pathogenesis of Alzheimer’s disease and stroke. Rat studies have shown inhibition of inflammatory cascades mitigate Alzheimer’s and stroke-related damage (8).
- Impaired neurogenesis – While most neurons are produced early in life, areas of the hippocampus and olfactory bulb continue to produce new neurons from stem cells in the adult brain. Reductions in this neurogenesis are a normal consequence of aging and may contribute to associated cognitive impairment (9).
Alterations in brain metabolism are a well-known element of neurodegenerative diseases, with deficiencies in glucose metabolism appearing early in the development of Alzheimer’s, prior to other symptoms (10). Parkinson’s patients similarly show reduced glucose metabolism in areas associated with motor control (11). Insulin resistance is associated with loss of cognitive function, and decreased glucose consumption/metabolism is seen in the temporal and parietal lobes of elderly subjects with cognitive impairment (12). This leads the review authors to argue metabolic defects may play a role in the causation and/or development of neurodegenerative disease, as summarized in Figures 3 and 4, below.
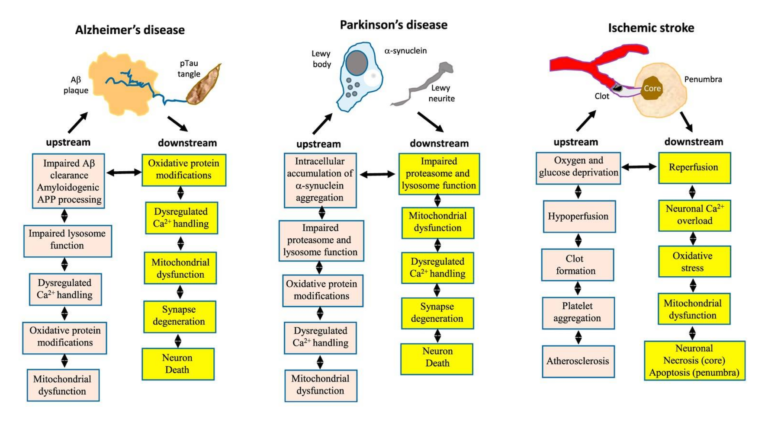
Figure 3: The hallmarks of brain aging likely contribute to or increase neuronal susceptibility to the physiological defects associated with Alzheimer’s, Parkinson’s, and stroke.
-
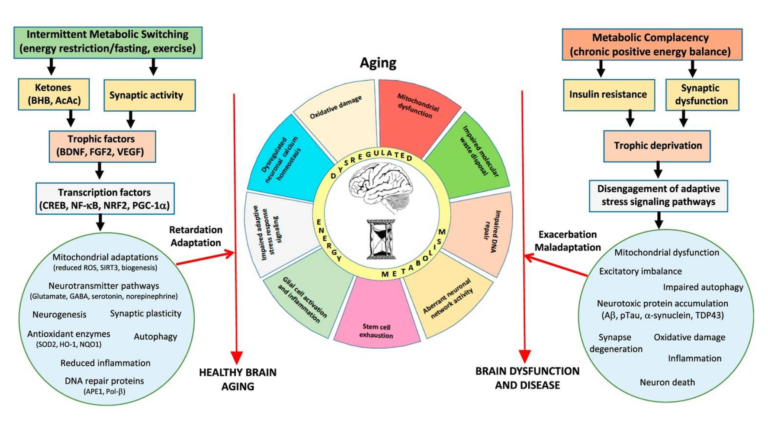
Figure 4: Intermittent depletion of liver glycogen stores and mobilization of fatty acids activate pathways that resist or reverse the consequences of aging, including “mitochondrial biogenesis and stress resistance; adaptive modifications of neurotransmitter signaling pathways; upregulation of autophagy, antioxidant defenses, and DNA repair; stimulation of neurogenesis; and suppression of inflammation.” Chronic positive energy balance, conversely, contributes to these same hallmarks and thus to aging, Alzheimer’s, Parkinson’s, and stroke.
For example, amyloid beta plaques are widely implicated as the cause of Alzheimer’s disease, but there are both significant elderly populations that have high amyloid levels but do not have Alzheimer’s and populations that have low amyloid levels and do suffer from the disease (13). The review authors argue amyloid accumulation may be a natural consequence of aging, and the hallmarks of aging outlined above create neuronal cells that are susceptible to amyloid-based damage. Similar mechanisms may link Parkinson’s and stroke to these same pathologies (14).
Poor metabolic health has been linked to poor cognitive performance, reduced gray matter, and reduced white matter integrity (15). Higher BMI is associated with reduced cerebral glucose utilization (16). In mice, insulin resistance is associated with increased brain inflammation, oxidative damage, poor calcium regulation, impaired learning and memory, and increased stress vulnerability (17).
Conversely, both exercise and intermittent energy restriction have been linked to improvements in mental and motor performance and robustness against neurodegenerative disease (18). As shown in Figure 4, the authors hypothesize these intermittent metabolic stressors upregulate cellular clearance and repair measures such as autophagy, DNA repair, and stress resistance and clearance; subsequent refeeding leads to protein production, cellular growth, and restoration (19). This is similar to the “metabolic switch” theory previously discussed on CrossFit.com and indicates intermittent bioenergetic challenges — challenges such as extended exercise or intermittent fasting, which lead to clearance of liver glycogen stores and a temporary reliance on circulating fats as a primary fuel source — repair and support resistance to the factors leading to brain aging and neurodegenerative disease.
Takeaway: Poor metabolic health can be linked, directly or indirectly, to the majority of the biological factors associated with brain aging and neurodegenerative disease. Mechanistic understanding and animal models suggest fasting and/or exercise can slow or reverse the progression of brain aging and disease.
Notes
- Mitochondria in neuroplasticity and neurological disorders; Mitohormesis, an antiaging paradigm; Adaptive responses of neuronal mitochondria to bioenergetic challenges: Roles in neuroplasticity and disease resistance
- A novel neurological phenotype in mice lacking mitochondrial manganese superoxide dismutase; Reduced mitochondrial SOD displays mortality characteristics reminiscent of natural aging
- TFEB overexpression in the P301S model of tauopathy mitigates increased PHF1 levels and lipofuscin puncta and rescues memory deficits; The role of autophagy in neurodegenerative disease; Life and death in the trash heap: The ubiquitin proteasome pathway and UCHL1 in brain aging, neurodegenerative disease and cerebral ischemia; Mitophagy and Alzheimer’s disease: Cellular and molecular mechanisms
- Upregulation of calcium/calmodulin-dependent protein kinase IV improves memory formation and rescues memory loss with aging; Reversal of aging related neuronal Ca2+ dysregulation and cognitive impairment by delivery of a transgene encoding FK506-binding protein 12.6/1b to the hippocampus; H. Evolutionary and functional perspectives on signaling from neuronal surface to nucleus; Elevated postsynaptic [Ca2+]i and L-type calcium channel activity in aged hippocampal neurons: relationship to impaired synaptic plasticity; Ca2+ regulation and gene expression in normal brain aging.; Early and simultaneous emergence of multiple hippocampal biomarkers of aging is mediated by Ca2+-induced Ca2+ release
- Regulation of glucose transporter 3 surface expression by the AMP-activated protein kinase mediates tolerance to glutamate excitation in neurons; Phosphorylation of the voltage-gated potassium channel Kv2.1 by AMP-activated protein kinase regulates membrane excitability; AMPK: sensing glucose as well as cellular energy status; Pathways towards and away from Alzheimer’s disease; New insights into brain BDNF function in normal aging and Alzheimer disease; The cholinergic system in aging and neuronal degeneration
- Perturbations of neural circuitry in aging, mild cognitive impairment, and Alzheimer’s disease; Reorganization of brain networks in aging: A review of functional connectivity studies; The aging motor system as a model for plastic changes of GABA-mediated intracortical inhibition and their behavioral relevance; Reduced GABA(A) receptor-mediated tonic inhibition in aged rat auditory thalamus; Molecular aspects of age-related cognitive decline: the role of GABA signaling; Frontal gamma-aminobutyric acid concentrations are associated with cognitive performance in older adults; Disconnected aging: cerebral white matter integrity and age-related differences in cognition
- Neurodegeneration in accelerated aging; Genomic integrity and the ageing brain; BDNF and exercise enhance neuronal DNA repair by stimulating CREB-mediated production of apurinic/apyrimidinic endonuclease 1; Mitochondrial and nuclear DNA-repair capacity of various brain regions in mouse is altered in an age-dependent manner
- Sirt3 protects dopaminergic neurons from mitochondrial oxidative stress; Complement and microglia mediate early synapse loss in Alzheimer mouse model; Extensive innate immune gene activation accompanies brain aging, increasing vulnerability to cognitive decline and neurodegeneration: a microarray study; Neuroprotection in stroke by complement inhibition and immunoglobulin therapy
- When neurogenesis encounters aging and disease; Adult neurogenesis in the mammalian brain: significant answers and significant questions
- CSF phosporylated TAU protein levels correlate with cerebral glucose metabolism assessed with PET in Alzheimer’s disease; Disrupted energy metabolism and neuronal circuit dysfunction in cognitive impairment and Alzheimer’s disease; Mitochondrial dysfunction and mitophagy in Parkinson’s: From familial to sporadic disease; Bioenergetics and metabolism: a bench to bedside perspective; Aberrant subcellular neuronal calcium regulation in aging and Alzheimer’s disease
- Networks mediating the clinical effects of pallidal brain stimulation for Parkinson’s disease: a PET study of resting-state glucose metabolism
- Brain fluorodeoxyglucose (FDG) PET in dementia; Impaired glucose tolerance in midlife and longitudinal changes in brain function during aging; Insulin resistance and Alzheimer’s disease: bioenergetic linkages
- Energy intake and exercise as determinants of brain health and vulnerability to injury and disease; Late-onset dementia: A mosaic of prototypical pathologies modifiable by diet and lifestyle; Comparison of neuropathologic criteria for the diagnosis of Alzheimer’s disease
- The etiology of Parkinson’s disease with emphasis on the MPTP story; Mitochondrial DNA and Parkinson’s disease; Trophic factors for Parkinson’s disease: To live or let die; How neuroinflammation contributes to neurodegeneration; Inefficient DNA repair is an aging-related modifier of Parkinson’s disease; Inhibitory dysfunction contributes to some of the motor and non-motor symptoms of movement disorders and psychiatric disorders, etc.
- Brain insulin resistance at the crossroads of metabolic and cognitive disorders in humans; Compromised white matter integrity in obesity; Body-brain connections: the effects of obesity and behavioral interventions on neurocognitive aging; Abdominal obesity and lower gray matter volume: a Mendelian randomization study; Association between waist circumference and gray matter volume in 2344 individuals from two adult community-based samples
- Inverse association between BMI and prefrontal metabolic activity in healthy adults
- Region-specific vulnerability to oxidative stress, neuroinflammation, and tau hyperphosphorylation in experimental diabetes mellitus mice; Diet-induced obesity and low testosterone increase neuroinflammation and impair neural function; Hippocampal calcium dysregulation at the nexus of diabetes and brain aging; Highfat diet impairs hippocampal neurogenesis in male rats; Diabetes impairs hippocampal function through glucocorticoid-mediated effects on new and mature neurons; Cognitive differences between Sprague-Dawley rats selectively bred for sensitivity or resistance to diet induced obesity
- Fasting: Molecular mechanisms and clinical applications; Metabolic control of longevity; Food restriction reduces brain damage and improves behavioral outcome following excitotoxic and metabolic insults; Dietary restriction normalizes glucose metabolism and BDNF levels, slows disease progression, and increases survival in huntingtin mutant mice; Dietary restriction and 2-deoxyglucose administration reduce focal ischemic brain damage and improve behavioral outcome: Evidence for a preconditioning mechanism; Intermittent fasting and caloric restriction ameliorate age-related behavioral deficits in the triple-transgenic mouse model of Alzheimer’s disease; Short-term fasting induces profound neuronal autophagy; Caloric restriction preserves memory and reduces anxiety of aging mice with early enhancement of neurovascular functions; Caloric restriction in older adults-differential effects of weight loss and reduced weight on brain structure and function
- The locus coeruleus: essential for maintaining cognitive function and the aging brain; Intermittent metabolic switching, neuroplasticity and brain health; Brain metabolism in health, aging, and neurodegeneration
Comments on Hallmarks of Brain Aging: Adaptive and Pathological Modification by Metabolic States
I stopped the PD medications prescribed due to severe side effects and started on natural treatments from VineHealth Center (VHC) in California, the herbal treatment has made a huge difference for me. My symptoms disappeared after the months long treatment! Go to ww w. vinehealthcenter. c om. This treatment is amazing!
Hallmarks of Brain Aging: Adaptive and Pathological Modification by Metabolic States
1