This 2017 paper reviews the potential impact of ketogenic diets on cancer.
As shown in Figure 1 below, many cancer cells metabolize glucose (the Warburg effect) through a process known as aerobic glycolysis. This unusual process may be used to create building blocks for cell replication or minimize the production of free radicals from normal glucose metabolism. This same process, however, generates little energy from each glucose molecule consumed and thus requires cancer cells to consume significantly more glucose to meet their energy needs. When glucose availability is restricted, cancer cells can be forced to use cellular respiration (glycolysis + oxidative phosphorylation), which simultaneously shifts the cell toward a non-proliferative state, thereby increasing oxidative damage and sensitivity to many cancer therapeutics. This process is summarized in Figure 1 below.
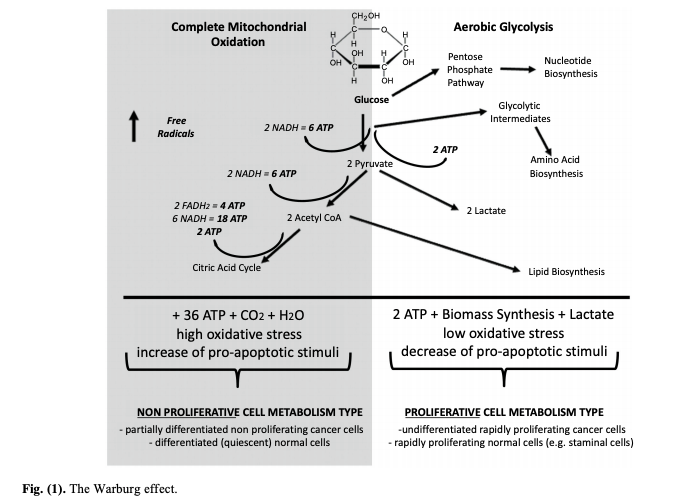
The paper also summarizes preliminary research on the use of ketogenic diets in cancer patients. The majority of this research has been performed on patients with glioblastoma multiforme (GBM), and early case reports have shown that the diet is tolerable and potentially extends patient lifespan. However, at the time of the paper’s publication, no controlled trial data existed comparing a ketogenic therapy — in addition to or separate from standard of care — to traditional treatments. With regard to other cancers, prospective studies suggest hormonal changes linked to carbohydrate restriction (including, but not limited to, suppression of insulin-related pathways) may reduce cancer risk or slow cancer progression.
The paper’s authors review a variety of potential mechanisms. Many of the direct downstream effects of glucose and/or insulin suppression reduce activation of pathways linked to tumor growth and proliferation. Additionally, a ketogenic diet may increase the effectiveness of the immune response to cancer cells. A consequence of aerobic glycolysis is the development of an acidic microenvironment around the tumor (due to lactate production), and this inhibits an effective immune response. In addition, immune cells, unlike many tumor cells, are capable of using ketones and other lipids as fuel. These cells thus remain healthy under the same metabolic conditions that increase tumor vulnerability.