Diabetes is associated with a variety of pulmonary defects including reduced expiratory volume (1), increased venous resistance (2) and changes to IGF-1 and ACE levels in the lungs (3). Diabetics are more susceptible to pulmonary infections (4). This 1997 hamster study sought to better understand the specific morphological changes of the lung associated with diabetes.
Hamsters were divided into three groups: those given streptozotocin (which induces diabetes), those given streptozotocin plus a high-cholesterol diet, and controls. Note that in hamsters, unlike in humans, excess cholesterol consumption induces a dramatic elevation in blood cholesterol levels. The hamsters were followed for 24 weeks.
After only six weeks of hyperglycemia, morphological changes were observed in the hamsters’ lung cells and microstructures. These changes included:
- Increased numbers of biosynthetic organelles (endoplasmic reticulum, Golgi apparatus)
- Reduced numbers of organelles involved in degradation (lysosomes)
- Increased adherence of macrophages to the alveolar lumen
- Thickening of the basal lamina between cells within the alveoli
- Increased extracellular matrix in the cellular interstitium (i.e., the area between cells)
- An uneven distribution of anionic charge throughout the cellular membranes of alveolar cells
These changes were associated with narrowing or collapse of 35% of capillaries and 30% of alveoli by the 18th week. Many of these changes are similar to those previously seen in the arterial, myocardial, and retinal endothelium of diabetics (5). Similar changes were seen in the hamsters that were both diabetic and hyperlipidemic, with the speed and degree of progression generally greater in the hyperlipidemic hamsters.
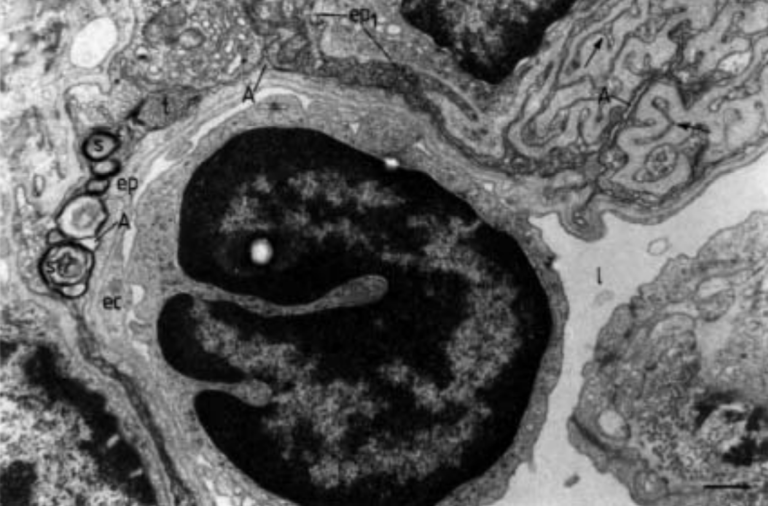
Fig. 5: Compressed lung alveolar space of a 24-week-old diabetic hamster. Over a large surface, the alveolar space (A) becomes collapsed, displaying compressed surfactant, which appears as tubular myelin (t) or as a multilayered structure (s). Note the osmiophilic type I epithelial cell (ep1), whose contorted contour (arrows) is accompanied by interstitial tissue (is), as well as the presence of apparently frequent collagen bundles (arrowheads), and elastin (e). l: vascular lumen; ec: endothelial cell; ep: type I epithelial cell. (Internal scale bar=0.430 µm).
Changes to the thickness and structure of the intracellular and extracellular matrices would be expected to impair alveolar gas transport. The uneven distribution of anionic charge would be expected to impair the adherence of circulating blood cells with the capillary lumen. Taken together, the authors conclude these changes contribute to impaired pulmonary function in the diabetic lung and indicate a direct link between diabetes and loss of lung function.
