Every 65 seconds, someone in the United States develops Alzheimer’s disease. It’s become so common that nearly one in 10 people over age 65 currently suffers from it, and, in a seemingly puzzling distribution, about two-thirds of those afflicted are women. In 2019, the ranks of Americans suffering with Alzheimer’s disease number 5.6 million older adults and 200,000 people younger than 40. In the next three decades, those numbers are expected to nearly triple and with them, the staggering cost in time and treasure, both in dollars and stolen lives.
The steep upward trajectory of the projected trend in the disease eerily mirrors that of the diabetes epidemic that began in the 1990s. Consider these images, both from the Centers for Disease Control; the inflection point for diabetes on the left — where the incidence began to sharply increase — occurred in the late 80s, about a decade after the introduction of the first USDA Food Guide Pyramid. The hockey stick jump for Alzheimer’s disease, according to the graph on the right (using data from 2000 through 2013), is projected to occur about now.
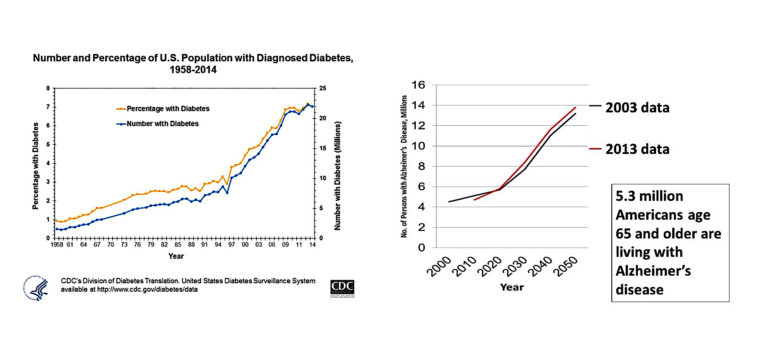
Little wonder, then, that researchers at Brown University coined the term “Type 3 diabetes” for Alzheimer’s disease.
But the reason for making that connection is based on more than the shape of the incidence curves. There is a fundamental and physiological link between defective glucose metabolism and brain dysfunction. People who suffer from diabetes, glucose intolerance, and insulin resistance (even early on, when blood sugar is still normal) have twice the risk of developing memory impairment as those without these disorders. And this connection has spurred deeper research into how the brain handles glucose and whether a defect in glucose metabolism within the brain affects the development of Alzheimer’s disease.
In one such avenue of research, scientists have used positron emission tomography (PET) scanning to examine the brains of people who carry the APOE-epsilon 4 gene (which confers a high risk of cardiovascular disease as well as a high risk of developing Alzheimer’s later in life) to determine the ability of different regions of the brain to use glucose. These studies unearthed a potentially important clue: The earliest indication of Alzheimer’s disease is a subtle decrease in the brain’s glucose metabolism, especially in the areas most closely tied to memory, and the changes can be identified in these individuals decades before memory starts to fail.
The brain is the most energy-expensive organ in the body, weighing only about three pounds but demanding the generation of between 300 and 400 kcal a day, which represents about 20% of the body’s resting energy expenditure. It cannot store fuel to any great degree either, so its function is almost totally dependent upon importing raw materials to produce the necessary energy. Fortunately, the brain comes with the flex-fuel option standard, so it’s able to run on either glucose or ketones. And though it actually runs more efficiently on ketones, in the vast majority of people eating the standard American diet (SAD) — high in starch, sugar, and industrially processed seed oils — glucose is the primary fuel, and some might say “drug,” of choice. The lion’s share of energy gobbled up by brains fed so SAD-ly comes from carbohydrate intake.
Glucose enters the brain and is ferried across the protective blood-brain barrier by specific transporters called GLUTs. Once across the barrier, various GLUTs also shuttle glucose into the brain cells, where it can be burned for energy. An interesting nexus of the female reproductive hormone estrogen and the GLUT system might explain the preponderance of females who develop Alzheimer’s disease. Estrogen stimulation drives the manufacture of these transporters, and its removal results in fewer of them. Diminished estrogen action equals fewer transporters, which equals less glucose transported for energy, which, as we’ll cover in a bit, sets the stage for brain dysfunction.
It was once believed that, unlike muscle and fat tissues, insulin was not required for glucose entry into brain cells, but research has since verified that insulin plays a number of critical roles, including involvement in the transmission of synaptic impulses, brain cell metabolism, memory, emotional and cognitive functions, and in the neuroinflammatory response. And where insulin plays a critical role (i.e., in a carb-filled world) can the specter of insulin resistance be far behind?
The brain requires a steady supply of incoming fuel to feed its energy needs. If glucose is the fuel option — which it will be on a highly processed, high-carb diet — insulin resistance is going to disrupt delivery of that steady supply. The brain cells will become relatively energy starved and, much like a rolling electrical brownout in the hot summer months, the lights will dim — i.e., the brain will operate but at a reduced capacity. Brain fog and memory dysfunction are a consequence, certainly, but that’s not even the worst of it.
A huge chunk of the energy the brain requires is devoted to running a bevy of intracellular pumps designed to handle the influx of excitatory brain chemicals, such as calcium ions and glutamate. The neuron needs these chemicals in the right amounts at the right time to fire impulses and process incoming stimuli on the one hand. On the other hand, these chemicals are, in a sense, toxic. These signaling molecules need to be let in to do their work and then actively pumped out. Each neuron expends great effort controlling the flow of these excitatory messengers, and when energy production dips, the pumps begin to fail, resulting in an excitatory chemical flood within the neuron. Left uncorrected, the energy-deprived and over-excited neurons can be damaged or even die. And since the functioning brain is just a collection of neurons, the ongoing attrition will ultimately take its toll.
Additionally, the development of insulin resistance in the brain and the attendant energy deficit may also play a key role in the development of the neurofibrillary tangles that are the signature lesion of the Alzheimer’s-affected brain. Neurons communicate with each other across specific small gaps between them called synapses. The development of a synapse occurs somewhat like the raising of a circus tent. The cell membrane can be envisioned as the floppy tent canvas, pushed up by tent poles (microfilaments) lashed together by ropes (tau proteins) that are glued securely together by a process (in the cell if not the circus) called phosphorylation. Without enough “glue,” the tent poles may come loose and fall; with too much glue they become a sticky, knotted mess — a tangle. The nexus of dysfunctional glucose metabolism, failing synapses, tau proteins, and tangles turns out to correspond to the degree of insulin resistance in the brain.
One solution for correcting insulin resistance in the body or the brain, and a simple one at that, is to flex the brain’s fuel preference. Flip the switch to select a more stable and predictable fuel source: fat in the form of ketones. Simply purging the system of highly processed, concentrated sources of sugar (and starch) and providing instead a rich intake of quality natural fats (short-, medium-, and long-chain triglycerides, primarily from animal sources) and sufficient complete protein to preserve lean tissues, supplemented with low-starch vegetables and some low-sugar fruits, will trigger a shift away from glucose dependency.
Additional Reading
- “A Different Perspective on the Treatment of Type 2 Diabetes”
- “A Different Perspective on the Treatment of Type 1 Diabetes”

Drs. Michael and Mary Dan Eades are the authors of 14 books in the fields of health, nutrition, and exercise, including the bestseller Protein Power.
Dr. Michael Eades was born in Springfield, Missouri, and educated in Missouri, Michigan, and California. He received his undergraduate degree in engineering from California State Polytechnic University and his medical degree from the University of Arkansas. After completing his medical and post-graduate training, he and his wife, Mary Dan, founded Medi-Stat Medical Clinics, a chain of ambulatory out-patient family care clinics in central Arkansas. Since 1986, Dr. Michael Eades has been in the full-time practice of bariatric, nutritional, and metabolic medicine. He and his wife have been in private practice devoting their clinical time exclusively to bariatric and nutritional medicine, gaining first-hand experience treating over 6,000 people suffering from high blood pressure, diabetes, elevated cholesterol and triglycerides, and obesity with their nutritional regimen.
Together, the Eades give numerous lectures to the general public and various lay organizations on their methods of treatment. They have both been guest nutritional experts on over 150 radio and television shows, including national segments for FOX and CBS.